Towards sensory Langmuir monolayers consisting of macrocyclic pentaaminoanthraquinone†
Abstract
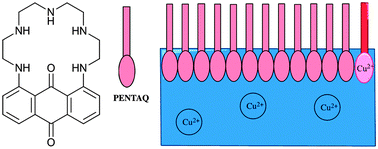
A pentaazamacrocycle incorporating an intracyclic anthraquinone fragment (PENTAQ) was synthesized with the aim of forming metal-responsive Langmuir monolayers. PENTAQ allows a good discrimination by naked eye of copper ions in methanol–water solutions (50 : 50 v/v). Spectrophotometric investigations of the protonation and Cu2+ binding properties of PENTAQ were undertaken in order to gain a deeper insight into the pH-dependent speciation as well as the color changing process. PENTAQ monolayers at air/water and air/methanol–water interfaces were prepared according to the Langmuir procedure. The structure of the monolayers can be tuned by varying the pH of the aqueous subphase, since it was found that the relative orientation at the interface of the aromatic lipophilic and polyazaalkyl hydrophilic fragments is critically dependent upon the protonation state of the immersed pentaazamacrocyclic chain. In situ fiber-optic UV-vis measurements revealed the formation of H-aggregates in the PENTAQ monolayer deposited on a water subphase at pH = 5.5. The ability of these films to detect copper(II) in the aqueous or methanol–water (20 : 80 v/v, pH = 9.8) subphase was explored by combining several experimental techniques, including pressure–area measurements (π–A isotherms), in situ UV-vis absorption spectrophotometry, and XPS spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...