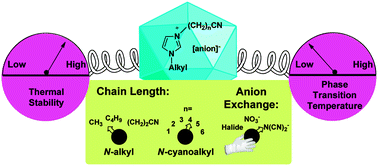
The synthesis of 10 N-alkyl-N-cyanoalkyl-functionalized imidazolium (N-methyl- and N-butyl-N-((CH2)nCN)imidazolium; n = 1–4) nitrate and 11 N-alkyl-N-cyanoalkyl-functionalized imidazolium (N-methyl-N-((CH2)nCN)imidazolium; n = 1–6, N-(2-cyanoethyl)-N-((CH2)nCN)imidazolium; n = 1,3–6) dicyanamide salts was achieved via N-alkylation of substituted imidazoles with commercially available haloalkylnitriles followed by anion exchange. Based on their observed melting points, all dicyanamide salts and all but one nitrate salt (1-cyanomethyl-3-methylimidazolium nitrate) had melting points <100 °C, as did 13 of the 17 halide precursors also reported here. Differential scanning calorimetry data indicated that melting points decreased by increasing the N-alkyl or N-cyanoalkyl chain length or by exchanging with the dicyanamide anion, which produced the lowest melting points in comparison to analogous halide or nitrate salts. Thermogravimetric analyses indicated that thermal stability increased for longer N-cyanoalkyl substituent lengths and decreased significantly for nitrates and more so for dicyanamides bearing short-chain N-cyanoalkyl substituents (e.g., N-cyanomethyl, N-(1-cyanoethyl), and N-(2-cyanoethyl)) in comparison to halide precursors. Furthermore, for many of the N-cyanoalkyl-substituted salts (especially the dicyanamides), there was a significant production of thermally-stable char – presumably due to by-products formed from the reaction of either N-cyanoalkyl substituents, dicyanamide anion, or both, which resulted in thermally-stable polymers or cycles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...