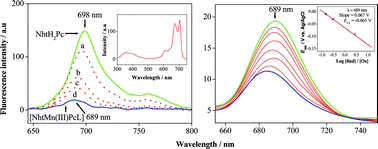
A new non-aggregating and electrochromic manganese(3+) phthalocyanine, (acetato)[2(3),9(10),16(17),23(24)-tetrakis(4-(1-naphthoxy-4-sulfonic acid sodium salt))phthalocyaninato]manganese(3+) [NhtMn(3+)PcL] (where Nht and L indicate naphthoxy-4-sulfonic acid sodium salt and acetate ion, respectively), was synthesized by reaction of the metal-free phthalocyanine (NhtH2Pc) and manganese(2+) acetate (Mn(OAc)2) in methanol. The formation of the complex was monitored by both in situUV-Vis and fluorescence techniques based on the change of the symmetry and the quenching of the fluorescence-probe molecule as a result of the insertion of the metal into the cavity of the macrocycle. The electro-spectrochemical behavior of [NhtMn(3+)PcL] was investigated with thin-layer UV-Vis and fluorescence spectroelectrochemical methods in DMSO solution. The electrochemical studies revealed that [NhtMn(3+)PcL] exhibited two reversible one-electron reductions which were assigned to Mn(2+)Pc(2–)/Mn(3+)Pc(2–) and Mn(2+)Pc(3–)/Mn(2+)Pc(2–) reversible couples based on the metal and phthalocyanine ring reductions, respectively. All redox processes were accompanied with a change of color from dark green to green and purple which are clearly monitored on the surface of the light transparent platinum gauze working electrode in the thin-layer cell. In situUV-Vis and fluorescence spectroelectrochemical methods were also applied to determine the E1/2 (half-wave potential) and n (number of electrons) of the first reduction process, both of which confirmed the reversible one-electron [Mn(3+)Pc(2–)L] + e → [Mn(2+)Pc(2–)L]–reduction process.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...