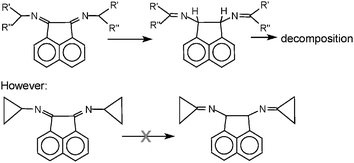
Ring strain is normally associated with increased reactivity and decreased stability of the strained molecule. However, we report here some examples in which the presence of a strained ring causes a stabilization of the molecule, allowing the isolation of some members of a class of otherwise unstable compounds. Alkyl-BIAN (alkyl-BIAN = bis(alkyl)acenaphthenequinonediimine) ligands have been elusive for 70 years. We have investigated the reason for earlier failures and identified it as an isomerization of the initially formed C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond. This isomerization is driven by a release of ring strain in the five-membered ring of the acenaphthene moiety. The use of amines in which the –NH2 group is bound to a quaternary carbon atom cannot be employed to avoid the isomerization because these amines are too sterically encumbered to react at all. However, the use of amines in which the amino group is bound to a strained ring solves the problem, because the isomerization would cause an even larger strain than the one that is released. Cyclopropylamine (Cypr-NH2) is the ideal amine, no isomerization being observed at all. Cyclobutylamine (Cybu-NH2) can also be employed, as well as amines in which the strain derives from the presence of a bi- o tri-cyclic system: 2-amino-exo-norbornane (Norb-NH2) and 2-aminoadamantane (Ad-NH2). The best synthetic procedure involves a transimination reaction from a [ZnCl2(Ar-BIAN)] complex, where Ar contains electron-withdrawing groups, but the direct synthesis from acenaphthenequinone and the amine is also possible in the case of Cypr-BIAN. The structure of [Pd(Cypr-BIAN)(η3-CH2C(CH3)CH2)][PF6], [ZnCl2(Cybu-BIAN)], [ZnCl2(Norb-BIAN)] and [NiBr2(Ad-BIAN)], has been determined by X-ray diffraction. Preliminary data indicate that Cypr-BIAN is a much stronger ligand than any Ar-BIAN compound.
N double bond. This isomerization is driven by a release of ring strain in the five-membered ring of the acenaphthene moiety. The use of amines in which the –NH2 group is bound to a quaternary carbon atom cannot be employed to avoid the isomerization because these amines are too sterically encumbered to react at all. However, the use of amines in which the amino group is bound to a strained ring solves the problem, because the isomerization would cause an even larger strain than the one that is released. Cyclopropylamine (Cypr-NH2) is the ideal amine, no isomerization being observed at all. Cyclobutylamine (Cybu-NH2) can also be employed, as well as amines in which the strain derives from the presence of a bi- o tri-cyclic system: 2-amino-exo-norbornane (Norb-NH2) and 2-aminoadamantane (Ad-NH2). The best synthetic procedure involves a transimination reaction from a [ZnCl2(Ar-BIAN)] complex, where Ar contains electron-withdrawing groups, but the direct synthesis from acenaphthenequinone and the amine is also possible in the case of Cypr-BIAN. The structure of [Pd(Cypr-BIAN)(η3-CH2C(CH3)CH2)][PF6], [ZnCl2(Cybu-BIAN)], [ZnCl2(Norb-BIAN)] and [NiBr2(Ad-BIAN)], has been determined by X-ray diffraction. Preliminary data indicate that Cypr-BIAN is a much stronger ligand than any Ar-BIAN compound.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond. This isomerization is driven by a release of ring strain in the five-membered ring of the acenaphthene moiety. The use of
N double bond. This isomerization is driven by a release of ring strain in the five-membered ring of the acenaphthene moiety. The use of 

 Please wait while we load your content...
Please wait while we load your content...