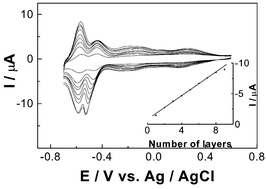
A novel sandwich-type compound, Na12[Fe4(H2O)2(As2W15O56)2]·41H2O, has been synthesized. The compound was well-characterized by means of IR, UV-vis, 183W NMR and elemental analyses. The compound crystallizes in the triclinic, Pī symmetry group. The structure of the compound is similar to that of Na16[M4(H2O)2(As2W15O56)2]·nH2O (M = Cu, Zn, Co, Ni, Mn, Cd), and consists of an oxo-aqua tetranuclear iron core, [FeIII4O14(H2O)2], sandwiched by two trivacant α-Wells-Dawson structural moieties, α-[As2W15O56]. Redox electrochemistry of the compound has been studied in buffer solutions at pH = 4.7 using polarography and cyclic voltammetry (CV). The compound exhibited four one-electron couples associated with the Fe(III) center followed by three four-electron redox processes attributed to the tungsten-oxo framework. The compound-containing monolayer and multilayer films have been fabricated on a 4-aminobenzoic acid modified glassy carbon electrode surface by alternating deposition with a quaternized poly(4-vinylpyridine) partially complexed with [Os(bpy)2Cl]2+/−. CV, X-ray photoelectron spectroscopy (XPS), UV-vis spectroscopy and atomic force microscopy (AFM) have been used to characterize the multilayer films. It is proved that the multilayer films are uniform and stable. The electrocatalytic activities of the multilayer films have been investigated on the reduction of two substrates of important analytical interest, NO2− and H2O2.

 Please wait while we load your content...
Please wait while we load your content...