Rational design of C2N-based type-II heterojunctions for overall photocatalytic water splitting†
Abstract
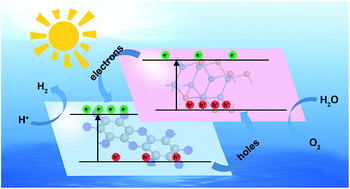
Photocatalytic water splitting is a promising method for the production of clean energy and searching for efficient photocatalysts has received extensive attention. Fabricating type-II heterojunctions is an effective approach to improve the photocatalytic efficiency. Based on the band edge positions and lattice parameters, we found that several kinds of monochalcogenide monolayers can be used to fabricate type-II heterojunctions with C2N monolayers. C2N/GaTe and C2N/InTe van der Waals (vdW) heterojunctions were investigated as potential photocatalysts for water splitting by means of first-principles computations. Both are type-II heterojunctions, and could promote the efficient spatial separation of electron–hole pairs. Their band edges straddle water redox potentials, satisfying the requirements for photocatalytic water splitting. Besides, the high carrier mobility of C2N/GaTe and C2N/InTe heterojunctions implies that the transfer of carriers to reactive sites is easy, and the recombination probability of photo-generated carriers is reduced. The Gibbs free energy calculations indicate that C2N/GaTe and C2N/InTe heterojunctions, especially C2N/InTe, exhibit high catalytic performance towards hydrogen and oxygen evolution reactions. Particularly, C2N/InTe exhibits a direct band gap with strong absorption in both visible and near ultraviolet regions, indicating that it is a very promising candidate for photocatalytic water splitting. This work would provide a new idea for the development of type-II heterojunctions for photocatalytic water splitting.
- This article is part of the themed collections: Editor’s Choice: Computational studies of nanomaterials for energy, catalysis and electronics and Photocatalysis and Photoelectrochemistry


 Please wait while we load your content...
Please wait while we load your content...