Non-coordinative metal selectivity bias in human metallothioneins metal–thiolate clusters†
Abstract
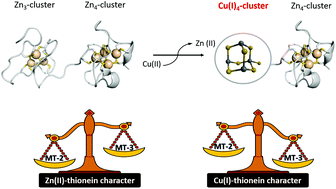
Mammalian metallothioneins (MT-1 through MT-4) are a class of metal binding proteins containing two metal–thiolate clusters formed through the preferential coordination of d10 metals, Cu(I) and Zn(II), by 20 conserved cysteine residues located in two protein domains. MT metalation (homometallic or heterometallic Zn(II)/Cu(I) species) appears to be isoform specific and controlling zinc and copper concentrations to perform specific and distinct biological functions. Structural and functional relationships, and in vivo metalation studies, identified evolutionary features defining the metal-selectivity nature for MTs. Metallothionein-3 (MT-3) has been shown to possess the most pronounced Cu–thionein character forming Cu(I)-containing species more favorably than metallothionein-2 (MT-2), which possesses the strongest Zn–thionein character. In this work, we identify isoform-specific determinants which control metal binding selectivity bias in different MTs isoforms. By studying the reactivity of Zn7MT-2, Zn7MT-3 and Zn7MT-3 mutants towards Cu(II) to form Cu(I)4Zn4MTs, we have identified isoform-specific key non-coordinating residues governing folding/outer sphere control of metal selectivity bias in MTs metal clusters. By mutating selected residues and motifs in MT-3 to the corresponding MT-2 amino acids, we dissected key roles in modulating cluster dynamic and metal exchange rates, in increasing the Cu(I)-affinity in MT-3 N-terminal β-domain and/or modulating the higher stability of the Zn(II)–thiolate cluster in MT-2 β-domain. We thus engineered MT-3 variants in which the copper–thionein character is converted into a zinc–thionein. These results provide new insights into the molecular determinants governing metal selectivity in metal–thiolate clusters.
- This article is part of the themed collection: In memory of Silvia Atrian


 Please wait while we load your content...
Please wait while we load your content...