A unifying view of the broad-spectrum antiviral activity of RSAD2 (viperin) based on its radical-SAM chemistry
Abstract
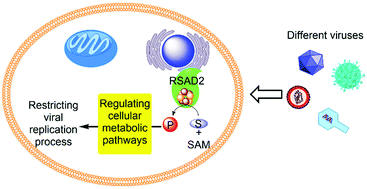
RSAD2 (cig-5), also known as viperin (virus inhibitory protein, endoplasmic reticulum associated, interferon inducible), is a member of the radical S-adenosylmethionine (SAM) superfamily of enzymes. Since the discovery of this enzyme more than a decade ago, numerous studies have shown that it exhibits antiviral activity against a wide range of viruses. However, there is no clear picture demonstrating the mechanism by which RSAD2 restricts the replication process of different viruses, largely because there is no direct evidence describing its in vivo enzymatic activity. As a result, a multifunctionality model has emerged. According to this model the mechanism by which RSAD2 restricts replication of different viruses varies and in many cases is not dependent on the radical-SAM chemistry of RSAD2. If the radical-SAM activity of RSAD2 is not required for its antiviral function, the question worth asking is: why does the cellular defence mechanism induce the expression of the radical-SAM enzyme RSAD2, which is metabolically expensive due to the requirement for a [4Fe–4S] cluster and usage of SAM? Here, in contrast to the multifunctionality view, I put forward a unifying model. I postulate that the radical-SAM activity of RSAD2 modulates cellular metabolic pathways essential for viral replication and/or cell proliferation and survival. As a result, its catalytic activity restricts the replication of a wide range of viruses via a common cellular function. This view is based on recent discoveries hinting towards possible substrates of RSAD2, re-evaluation of previous studies regarding the antiviral activity of RSAD2, and accumulating evidence suggesting a role of human RSAD2 in the metabolic reprogramming of cells.
- This article is part of the themed collection: Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...