Fe–oxy adducts of heme–Aβ and heme–hIAPP complexes: intermediates in ROS generation†
Abstract
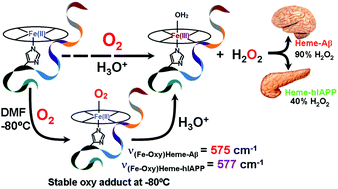
Iron (Fe) is the most abundant transition metal ion in the human body and its role, in the form of heme, has been implicated in Alzheimer's disease (AD) and type 2 diabetes mellitus (T2Dm). Heme binds both amyloid beta (Aβ) and human islet amyloid polypeptide (hIAPP) to form heme–Aβ and heme–hIAPP complexes, respectively, and form reactive oxygen species (ROS) like H2O2, O2˙−etc., which are known to cause oxidative damage. However the intermediates involved during ROS formation have not yet been isolated. In this study the oxygen bound intermediates of both heme–Aβ(1–16) and heme–hIAPP(1–19) have been isolated and characterized using absorption, EPR and resonance Raman (rR) spectroscopy. Fe–O stretches have been found at 575 cm−1 and 577 cm−1 for heme–Aβ(1–16) and heme–hIAPP(1–19) respectively. The oxy intermediates are stable at low temperatures. The isolation of the intermediates reveals a mechanistic pathway of ROS generation through the two heme complexes.

 Please wait while we load your content...
Please wait while we load your content...