Polymorphism controls the degree of charge transfer in a molecularly doped semiconducting polymer†
Abstract
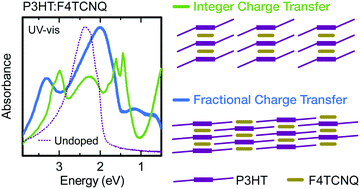
When an organic semiconductor (OSC) is blended with an electron acceptor molecule that can act as a p-type dopant, there should ideally be complete (integer) transfer of charge from the OSC to the dopant. However, some dopant–OSC blends instead form charge transfer complexes (CTCs), characterized by fractional charge transfer (CT) and strong orbital hybridization between the two molecules. Fractional CT doping does not efficiently generate free charge carriers, but it is unclear what conditions lead to incomplete charge transfer. Here we show that by modifying film processing conditions in the semiconductor–dopant couple poly(3-hexylthiophene):2,3,5,6-tetrafluoro-7,7,8,8,-tetracyanoquinodimethane (P3HT:F4TCNQ), we can selectively obtain nearly pure integer or fractional CT phases. Fractional CT films show electrical conductivities approximately 2 orders of magnitude lower than corresponding integer CT films, and remarkably different optical absorption spectra. Grazing incidence wide-angle X-ray diffraction (GIXD) reveals that fractional CT films display an unusually dense and well-ordered crystal structure. These films show lower paracrystallinity and shorter lamellar and π-stacking distances than undoped films processed under similar conditions. Using plane-wave DFT we obtain a structure with unit cell parameters closely matching those observed by GIXD. This first-ever observation of both fractional and integer CT in a single OSC–dopant system demonstrates the importance of structural effects on OSC doping and opens the door to further studies.
- This article is part of the themed collection: Materials Horizons Most Popular Articles


 Please wait while we load your content...
Please wait while we load your content...
