Origin of the high oxygen reduction reaction of nitrogen and sulfur co-doped MOF-derived nanocarbon electrocatalysts†
Abstract
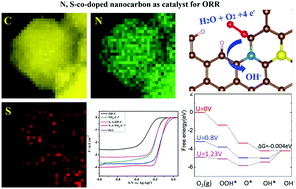
Developing an economical, highly active and durable material to replace the conventional, expensive noble metal electrocatalyst is an important milestone in the development of fuel cell technology. Nanocarbon materials are considered as promising catalysts toward the oxygen reduction reaction (ORR) in fuel cells, due to their reasonable balance between low-cost, long-life durability and high catalytic activity in alkaline media. In this work, we present the fabrication of N,S-co-doped nanocarbon derived from a metal-organic framework (MOF) precursor for use as an electrocatalyst towards ORR. High resolution transmission electron microscopy (HRTEM) mapping demonstrates the uniform distribution of N and S atoms into the nanocarbon skeleton. The nitrogen absorption–desorption isotherms indicate that the MOF-derived N,S-co-doped nanocarbon has a high specific surface area (2439.9 m2 g−1) and a porous structure. Importantly, the N,S-co-doped nanocarbon exhibits higher catalytic activity toward ORR, better long-term stability and methanol tolerance than commercial Pt/C catalyst. First-principles calculations demonstrate that the remarkable electrochemical properties of N,S-co-doped nanocarbon are mainly attributed to the synergistic effect from the N and S dopants. Moreover, for the first time, it is revealed that the N,S-coupled dopants in nanocarbon can create active sites with higher catalytic activity for ORR than the isolated N and S-dopants. This finding on the structure–performance relationship of the co-doped nanocarbon provides guidelines for the design of high performance electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...