Cation exchange for thin film lead iodide perovskite interconversion†
Abstract
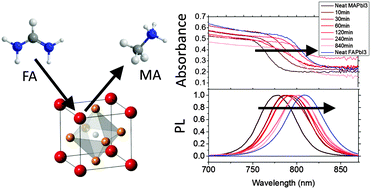
We report a new technique for tuning the bandgap of hybrid organic–inorganic halide perovskite materials. By dipping films of methylammonium or formamidinium lead triiodide (MAPbI3 or FAPbI3) in solutions of formamidinium or methylammonium iodide (FAI or MAI) at room temperature, we are able to inter-convert through cation exchange between perovskite materials, allowing us to carefully tune the bandgap between 1.57 and 1.48 eV. We observe uniform conversion through the entirety of the bulk film, with no evidence for a “bi-layered” or graded structure. By applying this technique to solar cell devices, we are able to enhance the performance of the single cation devices. Furthermore, we demonstrate that this technique allows us to form pure black phase FAPbI3 infiltrated into mesoporous scaffolds; this is normally infeasible since the pores confine the FAPbI3 in a yellow non-perovskite phase with a much wider bandgap, which is not of practical use in solar cells or other optoelectronic devices. Additionally, this work provides evidence for molecular cation mobility in the halide perovskites, indicating that the cations play a role in ionic conduction as well as the mobile anions.

 Please wait while we load your content...
Please wait while we load your content...