Development of a peptide-based bifunctional chelator conjugated to a cytotoxic drug for the treatment of melanotic melanoma†
Abstract
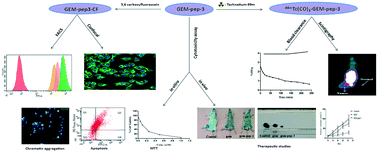
The cytotoxic drug gemcitabine (GEM) has been conjugated to receptor-binding peptides to target melanoma tumors. A hexapeptide having a Lys–Gly–His–Lys sequence (pep-1), an octapeptide with an Arg–Gly–Asp–Lys–Gly–His–Lys sequence (pep-2), a GEM-conjugated Lys–Gly–His–Lys peptide (GEM–pep-3) and a GEM-conjugated Asp–Gly–Arg peptide (GEM–pep-4) were synthesized and characterized. In vitro uptake of fluorescently labeled GEM–pep-3 and GEM–pep-4 on B16F10 cells was investigated. Fluorescence microscopy studies demonstrated significant uptake of GEM–pep-3 in the B16F10 mouse melanoma cell line. The peptides and GEM-coupled peptides were radiolabeled with [99mTc(CO)3(H2O)3]+ and examined for in vitro cell binding in the B16F10 melanoma cell line and in vivo biodistribution and scintigraphic studies in a B16F10 melanoma tumor-bearing mice model. In vitro cellular uptake studies and biological evaluation confirmed significant deposition of GEM–pep-3 at the melanoma tumor site. The MTT assay depicted higher cytotoxic behaviour of GEM–pep-3 than free GEM. A considerable amount of cell apoptosis was also observed in B16F10 cells. Finally, the in vivo therapeutic efficacy study revealed a significant decrease in tumor growth in the GEM–pep-3-treated animal model. These studies reveal enough potentiality of GEM–pep-3 to treat melanoma and underline the need for further evaluation.


 Please wait while we load your content...
Please wait while we load your content...