Design of a ligand for cancer imaging with long blood circulation and an enhanced accumulation ability in tumors†‡
Abstract
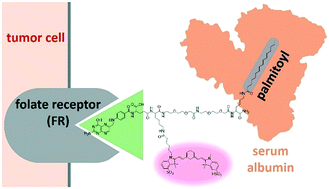
The use of imaging agents to visualize tumor cells is an advantageous technique to achieve a more efficient intraoperative diagnosis and effective debulking operations. Targeting of these agents to certain receptors that are overexpressed in cancer cells, such as the folate receptor, aids in tumor targeting. Several imaging probes have been developed using this strategy. However, these ligand-targeting cancer imaging probes are rapidly cleared during systemic delivery due to their small size, which compromises their biodistribution and circulation. Improving the detection of cancer requires higher accumulation and effective retention activities of imaging probes. Here we developed a new design for a folate-fluorophore conjugate that is modified with palmitoyl. Palmitic acid has a strong binding affinity with human serum albumin (HSA), which has the ability to form non-covalent host–guest complexes and has a blood half-life of 19 days. In this strategy, HSA is expected to serve as an endogenous nanocarrier for the designed probe in blood circulation. We hypothesized that via a reversible interaction with HSA, this simple palmitoyl modification on a folate–fluorophore conjugate can induce long blood circulation of the probe. Our folate-targeted probe could show longer blood circulation compared to the probe which lacks palmitoyl.


 Please wait while we load your content...
Please wait while we load your content...