Synthesis, DNA binding, docking and photocleavage studies of quinolinyl chalcones†
Abstract
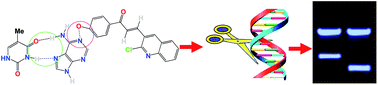
A series of simple quinoline–chalcone conjugates have been synthesized by Claisen–Schmidt condensation reactions of substituted acetophenones with 2-chloro-3-formyl-quinoline and evaluated for their nucleolytic activity. The structures of the synthesized quinoline–chalcone conjugates were confirmed by IR, 1H NMR, 13C NMR and mass spectral analyses. Most of the prepared compounds showed significant DNA binding and photocleavage activities. The incorporation of an electron-donating group into ring A caused a moderate increase in the DNA binding and photocleavage activities. Compounds 3c and 3d exhibited promising DNA photocleavage against pUC 19 DNA with 85% inhibition at 100 μM concentration. A structure–activity relationship analysis of these compounds was performed; compounds 3c and 3d are potential candidates for future drug discovery and development.

 Please wait while we load your content...
Please wait while we load your content...