Multiplexed end-point microfluidic chemotaxis assay using centrifugal alignment†
Abstract
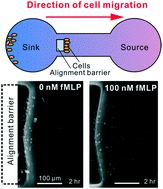
A fundamental challenge to multiplexing microfluidic chemotaxis assays at scale is the requirement for time-lapse imaging to continuously track migrating cells. Drug testing and drug screening applications require the ability to perform hundreds of experiments in parallel, which is not feasible for assays that require continuous imaging. To address this limitation, end-point chemotaxis assays have been developed using fluid flow to align cells in traps or sieves prior to cell migration. However, these methods require precisely controlled fluid flow to transport cells to the correct location without undesirable mechanical stress, which introduce significant set up time and design complexity. Here, we describe a microfluidic device that eliminates the need for precise flow control by using centrifugation to align cells at a common starting point. A chemoattractant gradient is then formed using passive diffusion prior to chemotaxis in an incubated environment. This approach provides a simple and scalable approach to multiplexed chemotaxis assays. Centrifugal alignment is also insensitive to cell geometry, enabling this approach to be compatible with primary cell samples that are often heterogeneous. We demonstrate the capability of this approach by assessing chemotaxis of primary neutrophils in response to an fMLP (N-formyl-met-leu-phe) gradient. Our results show that cell alignment by centrifugation offers a potential avenue to develop scalable end-point multiplexed microfluidic chemotaxis assays.


 Please wait while we load your content...
Please wait while we load your content...