A high-throughput microfluidic microphysiological system (PREDICT-96) to recapitulate hepatocyte function in dynamic, re-circulating flow conditions†
Abstract
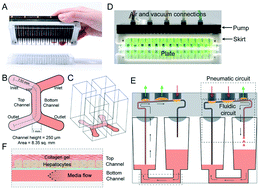
Microphysiological systems (MPSs) are dynamic cell culture systems that provide micro-environmental and external cues to support physiologically relevant, organ-specific functions. Recent progresses in MPS fabrication technologies have enabled the development of advanced models to capture microenvironments with physiological relevance, while increasing throughput and reducing material-based artefacts. In addition to conventional cell culture systems, advanced MPSs are emerging as ideal contenders for disease modeling and incorporation into drug screening. Since liver is a central organ for drug metabolism, liver-on-chip models have been developed to recapitulate hepatic microenvironment with varying complexities, while allowing long-term culture. Recently, we have developed a novel thermoplastic, oxygen-permeable MPS for primary human hepatocyte (PHH) culture. Herein, we have adapted and extended the MPS to a) a 96 microfluidic array (PREDICT-96 array) and b) integrated a novel, ultra-low volume, re-circulating pumping system (PREDICT-96 pump) – collectively known as the PREDICT-96 platform. The PREDICT-96 platform conforms to the industrial standard 96-well footprint and enables media re-circulation. First, we demonstrate the introduction of PHHs into the PREDICT-96 array using standard handling procedures for multi-well plates and allow cells to stabilize in static conditions. Next, we introduce recirculating flow into the bottom channel (using PREDICT-96 pump) to mimic mass transport in vivo. Our results indicate an increase in metabolic and secretory functions of PHHs in the PREDICT-96 platform, and their maintenance over 10 days of flow. Furthermore, long-term culture with fluid flow allows for the periodic introduction of media components (e.g., fatty acids, cytokines) and capture cellular responses to chronic stimuli. The low-volume footprint of the pump and small media volume in the MPS allow for the interrogation of hepatic responses incorporating secretion feedback to a stimulus, which is essential for disease model development and drug interrogation. We envision future development of this liver model to incorporate key primary hepatic cells, multi-cellular co-cultures and adaptation, integration with high-throughput analytical tools.
- This article is part of the themed collection: Organ-, body- and disease-on-a-chip systems


 Please wait while we load your content...
Please wait while we load your content...