High-throughput mechanotransduction in Drosophila embryos with mesofluidics†
Abstract
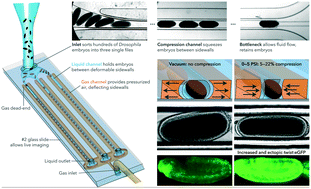
Developing embryos create complexity by expressing genes to coordinate movement which generates mechanical force. An emerging theory is that mechanical force can also serve as an input signal to regulate developmental gene expression. Experimental methods to apply mechanical stimulation to whole embryos have been limited, mainly to aspiration, indentation, or moving a coverslip; these approaches stimulate only a few embryos at a time and require manual alignment. A powerful approach for automation is microfluidic devices, which can precisely manipulate hundreds of samples. However, using microfluidics to apply mechanical stimulation has been limited to small cellular systems, with fewer applications for larger scale whole embryos. We developed a mesofluidic device that applies the precision and automation of microfluidics to the Drosophila embryo: high-throughput automatic alignment, immobilization, compression, real-time imaging, and recovery of hundreds of live embryos. We then use twist:eGFP embryos to show that the mechanical induction of twist depends on the dose and duration of compression. This device allows us to quantify responses to compression, map the distribution of ectopic twist, and measure embryo stiffness. For building mesofluidic devices, we describe modifications on ultra-thick photolithography, derive an analytical model that predicts the deflection of sidewalls, and discuss parametric calibration. This “mesomechanics” approach combines the high-throughput automation and precision of microfluidics with the biological relevance of live embryos to examine mechanotransduction. These analytical models facilitate the design of future devices to process multicellular organisms such as larvae, organoids, and mesoscale tissue samples.
- This article is part of the themed collection: Lab on a Chip Recent HOT Articles


 Please wait while we load your content...
Please wait while we load your content...
