Nitrogen-doped carbon nanotubes synthesized by pyrolysis of nitrogen-rich metal phthalocyanine derivatives for oxygen reduction†
Abstract
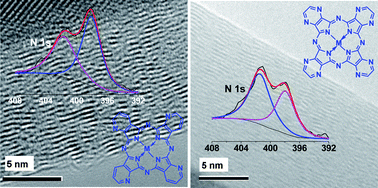
Two kinds of N-doped multiwalled carbon nanotubes (N-MWCNTs) S1 and S2 are prepared by pyrolysis of nitrogen-rich metal phthalocyanine derivatives, i.e. a mixture of metal tetrapyridinoporphyrazines (MTAPs, M = Fe2+, Ni2+) and tetrapyrazinoporphyrazines (MPTpzs) in a chemical vapour deposition furnace. The N-MWCNTs S1 and S2 have a straight structure and a high N/C atomic ratio. The ratios of N/C in S1 and S2 are 20.01% and 18.50%, respectively. The N atoms in S1 exhibit a uniform distribution, and the majority of N atoms are present in a pyridine type environment, while the N atoms in S2 are concentrated in certain areas, and are mainly in a graphite type environment. The electrocatalytic activities of the N-MWCNTs obtained are measured by the rotating disk electrode technique and cyclic voltammetry in an oxygen-saturated 0.1 M KOH solution. The results show that S1 exhibits a one-step, four-electron pathway for the reduction of oxygen (ORR), whereas S2 exhibits a two-step, two-electron process for ORR, S1 has much higher catalytic activity than S2.

 Please wait while we load your content...
Please wait while we load your content...