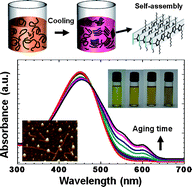
We demonstrated that interconnected nanofibrillar networks of poly(3-hexylthiophene) (P3HT) thin films with improved crystallinity can be easily fabricated by aging the precursor solution with marginal solvent. Structural analysis revealed that these benefits arise from the improvements in the crystallinity of P3HT in the precursor solution. At dilute concentrations, P3HT molecules grew into near-spherical particles during the aging time. As the aging time increased further, P3HT molecules exhibited one-dimensional growth into rod-like structures. At higher P3HT concentrations and longer P3HT solution aging times, dense nanowires were observed to form gradually, thereby improving the electronic properties of field-effect transistors (FETs) based on these films. This improvement was due to the change in P3HT organization in the precursor solution from a random-coil conformation to an ordered aggregate as a result of aging in a marginal solvent, methylene chloride. At high temperatures, the P3HT molecules were completely solvated and adopted a random-coil conformation, as is observed in good solvents. Whereas upon aging the solution at room temperature, methylene chloride poorly solvated the P3HT molecules such that ordered aggregates of P3HT grew in solution, which improved the molecular ordering of P3HT thin films produced from these solutions. The field-effect mobility of the thin films was, therefore, enhanced without the need for post-treatments.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...