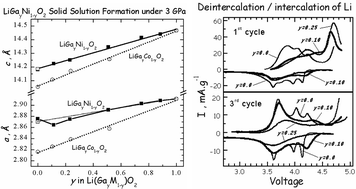
Solid solutions in the α-LiGaO2–LiNiO2 system were prepared over the whole concentration range under high pressure (3 GPa) at 700 °C in oxygen-rich atmosphere. The structure of LiGayNi1−yO2 solid solutions was characterized by XRD analysis, EPR and IR spectroscopy. It was found that Ga substitutes for Ni in the NiO2 layer (3a site), while Li and O are in their normal positions (3b and 6c, respectively). The occupancy of Ni in the Li site is negligible for Ga-substituted oxides. The progressive replacement of Ni by Ga leads to an unusual concentration variation of the a-parameter: at 10% Ga additives, there is a contraction of the a-parameter together with the expansion of the c-parameter. The concentration variation of the a-parameter can be related to increased trigonal distortion of GayNi1−yO2 layers upon Ga substitution. The electrochemical performance of LiGay(Ni1−xCox)1−yO2 solid solutions as a cathode material in lithium ion cells has been evaluated in potentiostatic experiments. During the first charge, there is an irreversible peak at about 4.7 V, which was compared with the electrochemical Li extraction from layered oxides characterized with a partial mixing of lithium and metal ions in the metal layers. After the first charge, a reversible electrochemical intercalation of Li is achieved. The effect of Ga on the potential of lithium extraction/insertion in LiGay(Ni1−xCox)1−yO2 was discussed.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...