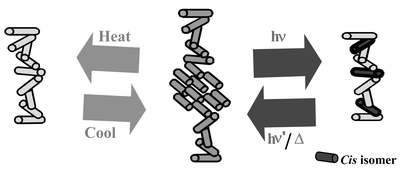
Thermal, photooptical as well as glass-forming properties of a series of photoresponsive chiral dimesogenic compounds containing cholesterol–azobenzene moieties linked to varying alkyl chain lengths (6–14) are described. All compounds except the dimesogen containing 7 methylene spacers show a cholesteric phase. Selective reflections observed in the cholesteric liquid crystals were found to be blue shifted upon increasing the temperature due to variations in smectic fluctuations. X-Ray diffraction studies of these compounds in the cholesteric phase show a low intensity reflection at small angles, the half widths of which decrease on decreasing the temperature, confirming the presence of smectic cybotactic groups. These materials also exhibit a strong odd–even effect in their reflection wavelength as a function of spacer length. Compounds with even numbers of methylene units show a drastic change in their reflection wavelength (102–225 nm) on temperature change compared to those with odd numbers of methylene units (9–14 nm). UV photolysis, using light of 366 nm wavelength, in the cholesteric super-cooled state exhibits a hypsochromic shift in the reflection wavelength. This can be explained by the formation of the cis isomer destroying the smectic clusters in the helical superstructure due to its bent geometry, which in turn causes a decrease in the reflected wavelength. A maximum wavelength shift of 215 nm (703–488 nm) has been observed on photolysis at 80 °C (cooling cycle) for the dimesogen having 12-methylene spacers. By rapid cooling of the compounds from the cholesteric temperature to 0 °C glassy liquid crystalline films maintaining helical molecular order were obtained. The optical properties of these dimesogens, such as cholesteric reflections, controlled by light and temperature combined with their glass forming properties, can be utilized for the construction of information storage materials.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...