Elucidation of the fate of zinc in model plants using single particle ICP-MS and ESI tandem MS†
Abstract
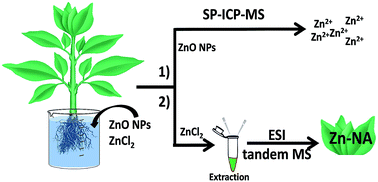
In recent years, the increasing use of zinc oxide nanoparticles (ZnO NPs) in many consumer products and industrial applications made them a new potential source of zinc in the environment. After their release into the environment, ZnO NPs can undergo different transformations which are still poorly understood and may influence their potential toxicity. This study investigates for the first time the fate of zinc, supplied in the form of ZnO NPs and ZnCl2, taken up by a model edible plant (lettuce, Lactuca sativa L.) by means of different mass spectrometry techniques. Single particle inductively coupled plasma mass spectrometry (SP-ICP-MS) analysis showed the rapid dissolution of ZnO NPs in the growth medium used for cultivation of lettuce, confirming that only dissolved zinc, not intact NPs, is taken up by plants. Taking into account that the beneficial or toxic effects of zinc depend on its form accumulated by plants, it is crucial to identify the chemical forms of zinc and their distribution in edible plant tissues. Therefore, the second part of this work was focused on the study of zinc speciation in plants cultivated with ZnCl2 by using hyphenated techniques. The proposed approach based on two-dimensional chromatographic (size exclusion – SEC and hydrophilic interaction – HILIC) separation with parallel ICP-MS and ESI-qTOF-MS/ESI-FT-Orbitrap-MS detection allows the determination and identification of minor zinc complexes at environmentally relevant concentrations. Nicotianamine (NA) is shown to be the major ligand binding zinc in lettuce leaves, and the Zn–NA complex is responsible for more than 70% of the total zinc content extracted from lettuce leaves by means of ammonium acetate buffer.
- This article is part of the themed collections: Community Leaders: Alfredo Sanz-Medel and JAAS Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...