Precise isotope analysis of sub-nanogram lead by total evaporation thermal ionization mass spectrometry (TE-TIMS) coupled with a 204Pb–207Pb double spike method†
Abstract
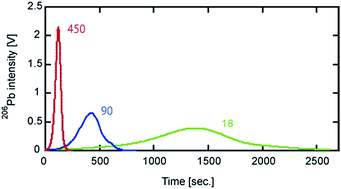
We present a new analytical technique for precise isotope measurement of sub-nanograms of Pb by total evaporation thermal ionization mass spectrometry (TE-TIMS) coupled with a 204Pb–207Pb double spike. The precise determination of Pb isotopic composition with a small (sub-nanograms) quantity of Pb is hampered due to the low signal intensity of 204Pb+, which has the lowest isotope abundance in natural samples (1.4%). The advantage of TE-TIMS is that the measured signal intensities are larger and the measurement time is shorter than those by the measurement with constant filament current. In this study, we investigated optimization of the analytical protocol for Pb isotope measurement by TE-TIMS coupled with a 204Pb–207Pb double spike, including the sample loading technique, filament temperature control, and the reduction method of isotope data acquired. The Pb isotope analysis was performed with the heating rate of 18, 90, and 450 mA min−1, with a short measurement time of 60, 30, and 20 minutes. The resulting reproducibilities of Pb isotope ratios were comparable to those of previous techniques including the 202Pb–205Pb double spike method and the 204Pb–207Pb double spike method using an amplifier with a state-of-the-art 1013 Ω resistor for collecting the 204Pb signal. The absolute Pb isotope ratios for NIST 981 measured by our method were biased due to anomalous 207Pb behavior occurring at the filament temperature of >1250 °C as well as the deterioration of Faraday cups. However, we confirmed that the accurate absolute Pb isotope ratios for an unknown sample can be obtained by normalizing the observed Pb isotope ratios to those of NIST 981 that are determined in the same analytical period and then multiplying the reference Pb isotope ratios of NIST 981. We measured the Pb isotope ratios of a standard rock material JB-3, of which the results were consistent with those reported by previous studies. We conclude that our method is suitable especially for the isotope analysis of sub-nanograms of Pb, and using a 204Pb–207Pb double spike instead of a 202Pb–205Pb double spike is beneficial for a number of laboratories due to the availability of the spikes. Furthermore, reduced measurement time compared to previous studies is helpful for measuring a large quantity of samples.


 Please wait while we load your content...
Please wait while we load your content...