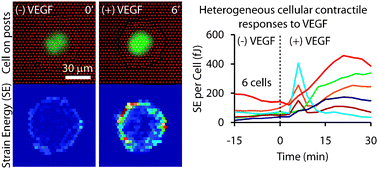
Mechanical traction forces exerted by adherent cells on their surroundings serve an important role in a multitude of cellular and physiological processes including cell motility and multicellular rearrangements. For endothelial cells, contraction also provides a means to disrupt cell–cell junctions during inflammation to increase permeability between blood and interstitial tissue compartments. The degree of contractility exhibited by endothelial cells is influenced by numerous soluble factors, such as thrombin, histamine, lysophosphatidic acid, sphingosine-1-phosphate, and vascular endothelial growth factor (VEGF). Upon binding to cell surface receptors, these agents trigger changes in cytoskeletal organization, adhesion and myosin II activity to varying degrees. While conventional antibody-based biochemical assays are suitable for detecting relatively large changes in biomarkers of contractility in an end-point format, they cannot resolve subtle or rapid changes in contractility and cannot do so noninvasively. To overcome these limitations, we developed an approach to measure the contractile response of single cells exposed to contractility agonists with high spatiotemporal resolution. A previously developed traction force sensor, comprised of dense arrays of elastomeric microposts on which cells are cultured, was combined with custom, semi-automated software developed here to extract strain energy measurements from thousands of time-lapse images of micropost arrays deformed by adherent cells. Using this approach we corroborated the differential effects of known agonists of contractility and characterized the dynamics of their effects. All of these agonists produced a characteristic first-order rise and plateau in forces, except VEGF, which stimulated an early transient spike in strain energy followed by a sustained increase. This novel, two-phase contractile response was present in a subpopulation of cells, was mediated through both VEGFR2 and ROCK activation, and its magnitude was modulated by receptor internalization. Interestingly, the concentration of VEGF could shift the proportion of cells that responded with a spike versus only a gradual increase in forces. Furthermore, cells repeatedly exposed to VEGF were found to contract with different dynamics after pretreatment, suggesting that exposure history can impact the mechanical response. These studies highlight the importance of direct measurements of traction force dynamics as a tool for studies of mechanotransduction.

 Please wait while we load your content...
Please wait while we load your content...