Energy-efficient butanol production by Clostridium acetobutylicum with histidine kinase knockouts to improve strain tolerance and process robustness†
Abstract
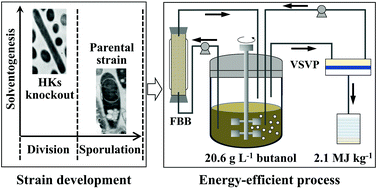
Under stress, Clostridium acetobutylicum sporulates and halts its metabolism, which limits its use in industrial acetone–butanol–ethanol (ABE) fermentation. It is challenging to manipulate the highly regulated sporulation program used by clostridia for survival. Four orphan histidine kinases (HKs) involved in the phosphorylation of Spo0A, a global regulator, were found to control sporulation, cell growth, and metabolism (acidogenesis and solventogenesis). Single knockout of cac3319 aborted sporulation at the initiation stage but enhanced butanol tolerance and production, whereas cac0437 knockout caused premature sporulation and inhibited the transition from acidogenesis to solventogenesis, resulting in early autolysis and low butanol production. No solvent was produced when both cac3319 and cac0903 were knocked out because the mutant lost the megaplasmid carrying solventogenesis genes. On the other hand, a mutant with double knockouts of cac3319 and cac0323 showed enhanced butanol production of >20 g L−1 by a repeated batch fermentation in a fibrous bed bioreactor, which enabled in situ product recovery via vapor stripping-vapor permeation to achieve the highest butanol production of 441.9 g L−1 ever reported. This study elucidated the regulatory roles of 4 HKs in controlling clostridial sporulation and solventogenesis with morphological, transcriptomics, and fermentation kinetics analyses, and demonstrated combinatory knockouts of HKs as a novel metabolic engineering approach to abort sporulation and enhance butanol production in ABE fermentation suitable for industrial application.


 Please wait while we load your content...
Please wait while we load your content...