Visible-light-induced denitrogenative phosphorylation of benzotriazinones: a metal- and additive-free method for accessing ortho-phosphorylated benzamide derivatives†
Abstract
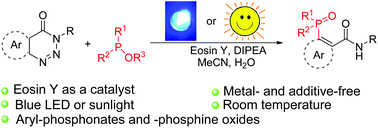
Metal-free, visible-light-induced denitrogenative phosphorylation of 1,2,3-benzotriazinones was achieved. With the use of eosin Y as a photoredox catalyst, N,N-diisopropylethylamine as a base, CH3CN–H2O as a solvent and sunlight or a blue LED as a light source, a variety of aryl-phosphonates, aryl-phosphinates, and aryl-phosphine oxides were efficiently prepared. In addition, B2pin2 instead of P-nucleophiles as a radical acceptor was also demonstrated. The key advantages of this newly developed method are the clean reaction profile, use of a low-cost organic-dye catalyst, energy efficiency, broad substrate scope, good to excellent yields and large-scale synthetic applicability. The gram-scale synthesised compounds could be isolated in pure form just upon extraction, followed by re-crystallisation; no tedious chromatographic purification was required.


 Please wait while we load your content...
Please wait while we load your content...