A novel method for screening deep eutectic solvent to recycle the cathode of Li-ion batteries†
Abstract
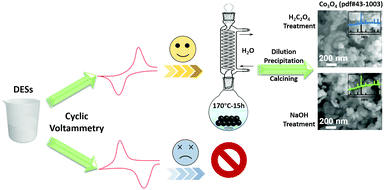
Deep eutectic solvents (DESs), as a green alternative technology, exhibit great potential to recycle valuable elements from spent lithium-ion batteries (LIBs). However, due to the weak reducing power of DESs, raising the temperature and extending the duration of processing are not efficient for screening suitable DESs for the recycling of LIBs. Here, we propose a novel, simple and robust experimental method to identify suitable DESs for recycling spent LIBs. Based on electrochemical principles, the method proposed in this study can be used to quickly determine the reducing power of DESs. As an example, a choline chloride (ChCl) and urea mixture as a DES was found to possess strong reducing power, with a Li and Co extraction efficiency of 95% from spent LIBs obtained at a reduced reaction temperature of 180 °C and reaction time of 12 h. The results are in good agreement with the results from Fukui functions calculations. The kinetic experiments revealed that the Li and Co extraction is controlled by solution and electron diffusion through the DES. Furthermore, a cubic cobalt oxide spinel (Co3O4) was obtained from the loaded DES using H2C2O4 and NaOH in the dilution–precipitation–calcination process. The current strategy demonstrates great potential for rapid and reliable screening of suitable DESs for the effective recycling of spent LIBs.
- This article is part of the themed collection: Battery science and technology – powered by chemistry


 Please wait while we load your content...
Please wait while we load your content...