Nano-V2O5/Ti porous membrane electrode with enhanced electrochemical activity for the high-efficiency oxidation of cyclohexane†
Abstract
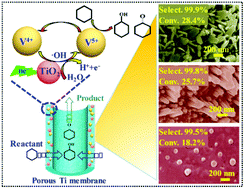
The selective oxidation of cyclohexane (CHA) is still a significant challenge in the field of current catalysis chemistry and chemical industry. Here, nano-V2O5/Ti porous membrane electrodes with different morphologies, i.e., nanorods (NRs), nanosheets (NSs), and nanoparticles (NPs), were prepared by loading nano-V2O5 onto porous Ti membranes. Simultaneously, a nano-V2O5/Ti membrane electrode was employed to constitute an electrocatalytic membrane reactor (ECMR) for CHA oxidation. Results indicated that the electrochemical performance was significantly associated with the morphology of the nano-V2O5 catalysts on the membrane electrode. ECMR with V2O5 NSs/Ti electrode exhibited superior properties. Excellent electrochemical activity was obtained in the application of CHA oxidation with 28.4% conversion and 99.9% selectivity to cyclohexanone and cyclohexanol (KA oil), which is better than most noble-metal-based catalysts reported in the literature under aerobic reaction conditions. Further, a prominent catalytic activity (10.5% conversion and 99.5% selectivity to KA oil) was first achieved even under 0 °C. The preferable electrocatalytic performance of V2O5 NSs/Ti electrode for CHA oxidation was attributed to the large effective electrocatalytic surface area, high electron transfer rate, and high rate of hydroxyl radical (˙OH) generation. Furthermore, the mechanism of CHA oxidation was explored. It was established that the valence change of V catalysts is an essential step during the selective oxidation of CHA by ECMR. Moreover, the V2O5 NSs/Ti electrode displayed excellent stability of oxidation during the selective oxidation of CHA. Therefore, our research opens up a new avenue toward the fundamental understanding and design of nanocatalysts in heterogeneous and electrochemical catalysis.


 Please wait while we load your content...
Please wait while we load your content...