High yield production of formate by hydrogenating CO2 derived ammonium carbamate/carbonate at room temperature†
Abstract
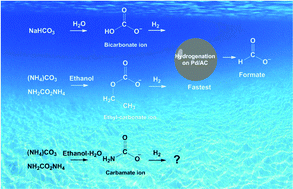
A sustainable economy is needed to recycle carbon rather than to irreversibly use stored carbon in fossil fuels. Instead of releasing CO2 into the atmosphere, its value can be recognized as a base C1 material for chemicals. In this study, a new CO2 utilization strategy is developed via the hydrogenation of CO2 derived ammonium carbamates/carbonates, which are the intermediates in industrial urea production or carbon capture processes, to produce value-added formate chemicals. A high yield of formate, ∼92%, was achieved after hydrogenating ammonium carbamates in the ethanol–water solution at room temperature with the carbon supported palladium nano-catalyst. The ethyl carbonate ions in the ammonium carbamate/carbonate solutions were speciated by 13C NMR and were attributed to the superior hydrogenation efficiency. Ammonium ions promote the formation of ethyl carbonate ions in the presence of ethanol. The solvent affects the distribution of the reactive intermediates.

 Please wait while we load your content...
Please wait while we load your content...