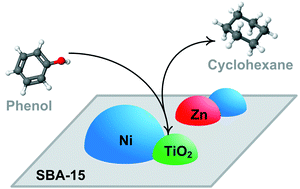
This work describes the effect of the support (TiO2, hybrid 2TiO2–SiO2, SBA-15 and SBA-15 decorated with TiO2 particles) on the catalytic activity of ZnNi catalysts in the gas-phase hydrodeoxygenation (HDO or O-removal) of phenol and the liquid-phase hydrodearomatization (HDA) of synthetic diesel. These reactions are representative of the two major challenges of the hydrotreating unit embedded in a sustainable refinery: (i) decreasing oxygen content of bio-oils (produced in the pyrolysis of lignocellulosic biomass); and (ii) decreasing aromatics content in diesel. The fresh and deactivated catalysts were characterized by XRD, N2 adsorption–desorption, TPR, MS/TPD-NH3, XPS, SEM, HRTEM and coke combustion. Under steady-state conditions, the ZnNi catalyst supported on SBA-15 decorated with TiO2 particles displayed the highest activity in the hydrodeoxygenation of phenol (selectivity toward deoxygenated products > 95%) whereas the ZnNi/SBA-15 catalyst displayed the highest activity in the hydrodearomatization of synthetic diesel. It has been shown that dispersion of the active ingredient is favoured on the SBA-15 substrate. The relationship between structure and activity demonstrated that HDO and HDA reactions require optimized metal dispersion and acid function, metal dispersion being more important for HDA than for HDO reactions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...