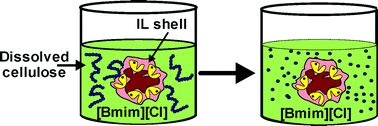
A new approach to improve the cellulase stability against 1-butyl-3-methylimidazolium chloride ([Bmim][Cl]), based on coating of immobilized enzyme particles with hydrophobic ILs, is proposed. The stability of commercial cellulase (Celluclast®), immobilized onto a polymeric support (Amberlite XAD4), was first studied in ten different ionic liquids (ILs) at 50 and 80 °C. Hydrophobic ILs clearly enhanced the enzyme thermal stability. Butyltrimethyl-ammonium bis(trifluoromethylsulfonyl)imide ([N1114][NTf2]) enhances half-life time of the immobilized enzyme at 50 °C up to 4 times, while [Bmim][Cl] behaved as a powerful enzyme deactivating agent. Thus, the stability of cellulase in hydrophobic IL/[Bmim][Cl] mixtures was greatly improved with respect to [Bmim][Cl] alone. A stabilized cellulase derivative obtained by coating immobilized enzyme particles with [N1114][NTf2], has been then successfully used for the saccharification of dissolved cellulose in [Bmim][Cl] (i.e. up to 50% hydrolysis in 24 h) at 50 °C and 1.5 w/v water content.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...