Enzymatic esterification in ionic liquids integrated with pervaporation for water removal†
Abstract
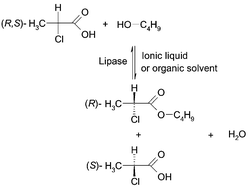
The application of ionic liquids as solvents for the enantioselective esterification of (R,S)-2-chloropropanoic acid with butan-1-ol using Candida rugosa lipase is reported. The role of water produced during the reaction and controlling of the water activity with pervaporation was studied. The enantioselective esterification of (R,S)-2-chloropropanoic acid with butan-1-ol in different organic solvents and ionic liquids was studied. The reaction was effectively catalysed by Candida rugosa lipase. From the ionic solvents investigated, the best results were achieved in [bmim]PF6. The presence of water had a strong effect on the activity of the lipase. Since in esterification reactions with acid and alcohol equimolar amounts of water are formed, the excess of water was removed and the water activity was kept constant using a pervaporation system without any additives. At the optimum water activity Candida rugosa lipase showed high thermal stability in [bmim]PF6 and it could be reused for at least five recycles with only a small lost of activity.

 Please wait while we load your content...
Please wait while we load your content...