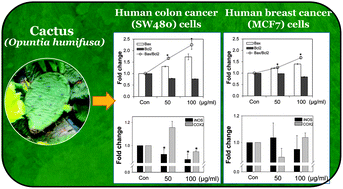
Cactus (Opuntia spp) is widely cultivated as a vegetable, fruit, and forage crop and has been used in traditional medicine in American Indian, Mexican, and Korean cultures. Accumulative evidence from both in vitro and in vivo studies using cacti suggests their biological and pharmacological activities, such as their anti-cancer and anti-inflammatory roles in different cancer cells. In this study, the Opuntia humifusa stem (OHS) was extracted with different solvents and screened for radical scavenging activity using 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS˙+) and 1,1-diphenyl-2-picryl hydrazyl (DPPH). In addition, the total phenolic and flavonoid contents of each extract were analyzed using the Folin–Ciocalteu method and high performance liquid chromatography, respectively. Further, the cacti's bioactive fractions were evaluated for cell cytotoxicity and to understand their mechanism of action on human colon cancer (SW480) and breast cancer (MCF7) cells. An ethyl acetate (EtOAc) extract exhibited the highest cytotoxicity and resulted in an up-regulated expression of the pro-apoptotic protein Bax (bcl-2 associated X protein) and a down-regulated expression of the anti-apoptotic protein Bcl2 in both SW480 and MCF7 cells. The apoptosis was mediated through activation of caspase 8, 9, and 3/7 activities as well as PARP cleavage in SW480 cells, while the same extract activated only a caspase 9 activity in MCF7 cells. Furthermore, incubation of cells with the EtOAc extract down-regulated the expression of inflammatory molecules such as cyclooxygenase-2 (COX2) and inducible nitric oxide synthase (iNOS) in SW480 cells but not in MCF7 cells. Taken together, these results suggest that SW480 colon cancer cells are more susceptible to bioactive compounds present in OHS and may have potential in the prevention of cancer through modulation of apoptosis markers and inhibition of inflammatory pathways.

 Please wait while we load your content...
Please wait while we load your content...