In vitro protein digestibility and physico-chemical properties of flours and protein concentrates from two varieties of lentil (Lens culinaris)
Abstract
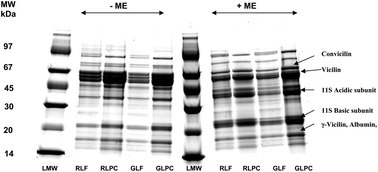
The chemical composition of whole lentil flours and lentil protein concentrates prepared by alkaline extraction and iso-electric precipitation from Blaze and Laird varieties of lentil were studied. The protein composition of the flours and concentrates, determined by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and size-exclusion high-performance liquid chromatography (SE-HPLC) showed that the extracted proteins were composed mainly of globulins and albumins. Trypsin inhibitor activity ranged between 0.94 and 1.94 trypsin inhibitor units (TIU) mg−1 for the flours, but was markedly lower in the protein concentrates ranging between 0.17 and 0.66 TIU mg−1. In vitro protein digestibility ranged between 75.90 and 77.05% for the flours, whereas significantly (P < 0.05) higher values, ∼82.80 to 83.20%, were determined for the concentrates. Significant (P < 0.05) differences in colour (ΔE) were observed between the flours and the concentrates from both varieties. Thermal properties of both flours as studied by differential scanning calorimetry (DSC) were comparable. However, the endothermic parameters of the two protein concentrates were significantly (P < 0.05) different. Overall, the results show that in vitro protein digestibility of lentil protein concentrates is higher than that of the flours, however, both lentil flours and protein concentrates contain useful proteins that could serve as value-added ingredients in food formulations.

 Please wait while we load your content...
Please wait while we load your content...