Photoinduced formation mechanism of the thymine–thymine (6–4) adduct in DNA; a QM(CASPT2//CASSCF):MM(AMBER) study†
Abstract
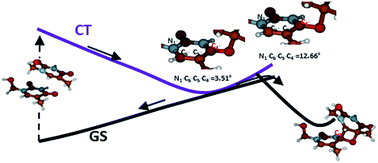
The UVB-induced photomechanism leading the carbonyl group of a thymine nucleobase to react with the carbon–carbon double bond of a consecutive thymine nucleobase in a DNA strand to form the thymine–thymine (6–4) photodamage adduct remains poorly understood. Key questions remain unanswered, concerning both the intrinsic features of the photoreaction (such as the contribution (or not) of triplet states, the nature of the involved states and the time-scale of the photoprocess) and the role played by the non-reactive surroundings of the two reactive pyrimidine nucleobases (such as the nature of the flanked nucleobases and the flexibility of the whole DNA molecule). A small number of theoretical studies have been carried out on the title photoreaction, most of which have used reduced model systems of DNA, consequently neglecting potential key parameters for the photoreaction such as the constraints due to the double strain structure and the presence of paired and stacked nucleobases. In the present contribution the photoactivation step of the title reaction has been studied in a DNA system, and in particular for a specific DNA hairpin for which the quantum yield of photodamage formation has been recently experimentally measured. The reaction has been characterized by carrying out high-level QM/MM computations, combining the CASPT2//CASSCF approach for the study of the reactive part (i.e. the two thymine molecules) with an MM-Amber treatment of the surrounding environment. The possibility of a reaction path along both the singlet and triplet manifolds has been characterized, the nature of the reactive states has been analyzed, and the role played by the flexibility of the whole system, which in turn determines the initial accessible geometrical conformations, has been evaluated, thus substantially contributing towards the elucidation of the photoreaction mechanism. On the basis of the obtained results, it can be observed that a charge-transfer state can decay from a pro-reactive initial structure towards a region of energy degeneracy with the ground state, from which the subsequent decay along the ground state hypersurface can lead to the photoreaction.
- This article is part of the themed collection: Photoinduced Processes in Nucleic Acids and Proteins


 Please wait while we load your content...
Please wait while we load your content...