Improved sludge dewaterability using persulfate activated by humic acid supported nanoscale zero-valent iron: effect on sludge characteristics and reaction mechanisms
Abstract
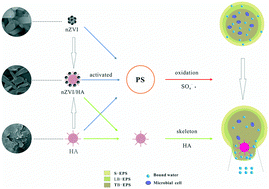
In this study, nanoscale zero-valent iron (nZVI) supported on humic acid (HA) as a novel nanocomposite (nZVI/HA) was synthesized and characterized by field emission scanning electron microscopy (FE-SEM), energy dispersive spectrometry (EDS), Fourier transform infrared (FTIR) spectroscopy, and the Brunauer–Emmett–Teller method. The nZVI/HA nanocomposite was used as a persulfate (PS) activator to enhance sludge dewatering, and the specific resistance to filterability (SRF) of sludge decreased from 2.75 × 1013 m kg−1 to 0.217 × 1013 m kg−1 after conditioning. Subsequently, variation in extracellular polymeric substances (EPS) under chemical conditioning was investigated concretely to reveal the reaction mechanisms. Analysis of the three-dimensional excitation emission matrix (3D-EEM) revealed that the reduction of the aromatic protein and tryptophan-like protein content in tightly-bound extracellular polymeric substances (TB-EPS) is conducive to the enhancement of sludge dewaterability. In addition, the introduction of HA particles effectively decreased the sludge compressibility relative to that of raw sludge by forming skeleton structures. The observed sludge microstructure properties clearly indicated that the treated sludge floc induced the formation of additional pore structures and water channels.


 Please wait while we load your content...
Please wait while we load your content...