Photo-oxidation of 11-nor-9-carboxy-Δ9-tetrahydrocannabinol using medium-pressure UV and UV/H2O2 – a kinetic study†
Abstract
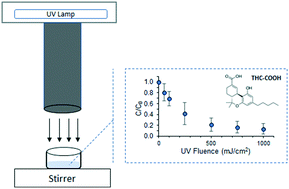
The main psychoactive compound in Cannabis sativa, Δ9-tetrahydrocannabinol (THC), and its metabolites are emerging organic contaminants that have been detected in waste and surface waters around the globe. This study tested the efficacy of advanced oxidation processes (i.e., direct UV photolysis or UV/H2O2 processes) towards the treatment of the main human metabolite of THC, THC-COOH. Three different waters were selected to evaluate the photolysis and oxidation performance with polychromatic UV light in the range of 200–300 nm, and the effect of pH on the photooxidation of THC-COOH was also studied. The results indicated that direct UV photolysis followed pseudo first-order kinetics, with fluence-based rate constants ranging from 1.9(0.3)–2.7(0.2) × 10−3 cm2 mJ−1, 0.6(0.2)–1.0(0.1) × 10−3 cm2 mJ−1, and 0.6(0.2)–0.9(0.2) × 10−3 cm2 mJ−1 for THC-COOH prepared in deionised (DI) water, Suwannee River humic acid (SRHA) dissolved in DI water, and natural lake water, respectively. This study found that the UV/H2O2 process did not enhance the oxidation rate in comparison to direct photolysis, and no effect of pH from 5.6 ± 0.2 to 8.7 ± 0.2 was observed. Hence, the hydroxyl radical (HO˙) degradation pathway played a less important role in the overall oxidation of THC-COOH under the tested conditions (i.e., SRHA and lake water) than direct photolysis.
- This article is part of the themed collection: Ultraviolet-based Advanced Oxidation Processes (UV AOPs)


 Please wait while we load your content...
Please wait while we load your content...