Emerging investigators series: comparative study of naproxen degradation by the UV/chlorine and the UV/H2O2 advanced oxidation processes†
Abstract
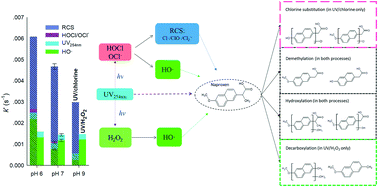
The UV/chlorine advanced oxidation process (AOP), which forms HO˙ and reactive chlorine species (RCS such as Cl˙, ClO˙ and Cl2˙−), is being considered as an alternative to the UV/H2O2 AOP for the degradation of emerging organic contaminants. This study compared the kinetics and pathways of the degradation of a recalcitrant pharmaceutically active compound, naproxen (NPX), by the UV/chlorine and UV/H2O2 AOPs. The degradation of NPX by both AOPs followed pseudo first-order kinetics, and, at pH 7, the first-order rate constant (k′) in UV/chlorine was 4.9 times higher than that in UV/H2O2. At pH 7, in the UV/chlorine process, the HO˙ and RCS contributed to 15.9% and 76.3%, respectively, of the NPX degradation. Radical scavenging tests indicated that ClO˙ and CO3˙− were important to the NPX degradation by UV/chlorine. A higher efficiency was observed in UV/chlorine than in UV/H2O2 at the pH range of 6–9, but as the pH rose from 6 to 9, k′ decreased from 6.10 × 10−3 s−1 to 2.98 × 10−3 s−1 in UV/chlorine. However, in UV/H2O2, k′ was only slightly affected by pH. In both AOPs, k′ increased linearly with increasing dosages of oxidants (chlorine or H2O2) from 20 μM to 200 μM. The UV/H2O2 process was less affected by the water matrix than the UV/chlorine process. Compared to pure water, k′ in tap water was reduced by 9% and 23.2% by UV/H2O2 and UV/chlorine, respectively. The degradation by both AOPs was associated with hydroxylation and demethylation. Decarboxylation was particularly observed in UV/H2O2, and chlorine substitution was observed in UV/chlorine. During the UV/chlorine process, the acute toxicity to Vibrio fischeri increased and then decreased in the system.
- This article is part of the themed collections: Emerging Investigator Series and Ultraviolet-based Advanced Oxidation Processes (UV AOPs)


 Please wait while we load your content...
Please wait while we load your content...