Emerging investigator series: significantly enhanced uptake of Eu3+ on a nanoporous zeolitic mineral in the presence of UO22+: insights into the impact of cation–cation interaction on the geochemical behavior of lanthanides and actinides†
Abstract
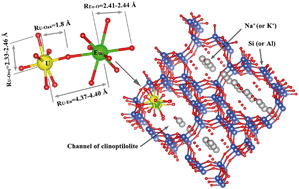
It is traditional understanding that different metal cations compete with each other for the sorption sites in a sorbent through surface complexation or ion exchange. For most natural minerals, two types of metal cations may suppress the uptake of each other when they coexist in solution. In this study, we report an abnormal co-sorption behavior of lanthanide cation Eu3+ and actinide cation UO22+ on clinoptilolite, a well-known zeolite that has been used in metal wastewater remediation. Batch experiments reveal that although the presence of Eu3+ does not affect UO22+ sorption on clinoptilolite, Eu3+ sorption is significantly enhanced by the presence of UO22+ while the degree of enhancement is highly dependent on UO22+ concentration. Extended X-ray absorption fine structure spectroscopy (EXAFS) and luminescence spectroscopy indicate that this unexpected enhancement originates from the further bridging of axial “yl” oxo atoms from uranyl cations to Eu3+ cations, traditionally known as cation–cation interactions (CCIs), a special type of bonding that was intensively studied in solution chemistry and solid state chemistry of actinyl cations, but has never been reported for environmental systems. This observation sheds light on better understanding of the chemical behavior of lanthanides and actinides during nuclear waste management and their fate prediction in the environment.
- This article is part of the themed collection: Emerging Investigators Series


 Please wait while we load your content...
Please wait while we load your content...