Enhanced heterogeneous activation of peroxymonosulfate by Co and N codoped porous carbon for degradation of organic pollutants: the synergism between Co and N†
Abstract
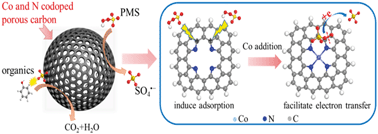
Rational design of high-efficiency carbon-based nanomaterials towards heterogeneous activation of peroxymonosulfate (PMS) for environmental remediation is highly desirable. Codoping of Co and N into carbon may synergistically modify the electronic structure of carbon and enhance its catalytic activity towards PMS. However, the synergistic mechanism between Co and N, especially the role of Co, remains unknown. Herein, Co and N codoped porous carbons (Co–N-PCs) were constructed for enhanced PMS activation. The Co doping level was varied to study the role of Co in Co–N-PCs. Results showed that codoping of Co and N into carbon generated a synergistic effect to enhance PMS activation for the degradation of organic pollutants. The kinetic constant of phenol degradation of the Co–N-PC was 48.4, or 17.6 times larger than that of porous carbon with only N or Co doping, and was superior to that of homogeneous Co2+ (best reported PMS catalyst). Moreover, the catalytic activity of Co–N-PC was significantly enhanced with the increase of Co–N coordination sites (Co–Nx), which could also inhibit Co leaching due to its strong binding. The experimental and DFT results revealed that the synergism of Co and N was ascribed to the combination of their different roles: the N doping mainly produced the charged carbon sites for PMS adsorption, while the Co doping mainly facilitated the electron transfer from carbon to PMS for its dissociation into sulfate radical.


 Please wait while we load your content...
Please wait while we load your content...