An oxygen-blocking oriented multifunctional solid–electrolyte interphase as a protective layer for a lithium metal anode in lithium–oxygen batteries†
Abstract
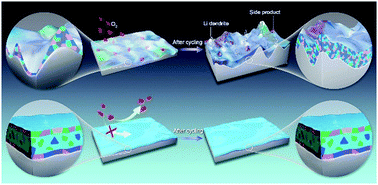
For a period of time, the O2 cathode has been the focus of research, while the study of lithium anodes and their interactions is quite rare in rechargeable lithium–oxygen (Li–O2) batteries. Actually, the unstable interface between a Li metal anode and the electrolyte in the presence of O2 will eventually lead to battery failure. It was proposed that lithium nitrate (LiNO3), a well-known stabilizing additive being able to form a passivation solid–electrolyte interphase (SEI) on the Li metal surface, could be introduced into the electrolyte of Li–O2 batteries to improve their performance. Nevertheless, the effective utilization of LiNO3 in this system has heretofore been limited due to the dissolution of NO2− species, one of the components of the SEI, as well as O2 corrosion issues on the Li metal side, thus resulting in unstable cycling of Li–O2 batteries. Herein, by combining the electrochemical polishing technology with the LiNO3 reduction chemistry, we construct on a Li metal surface a stable molecularly smooth SEI which possesses a unique multi-layered structure that encapsulates the soluble NO2− species inside the inner layer. Such an SEI film can not only avoid the negative effects caused by the dissolution of NO2−, but also effectively suppress the dendrite growth on and the O2 permeation to the Li surface. Consequently, the cycle lives of O2-saturated symmetric Li cells and Li–O2 batteries are both improved substantially. Our work offers an effective strategy to protect Li metal anodes in Li–O2 batteries, and meantime, provides a novel insight into the rational utilization of electrolyte additives.


 Please wait while we load your content...
Please wait while we load your content...