Optimizing mesostructured silver catalysts for selective carbon dioxide conversion into fuels†
Abstract
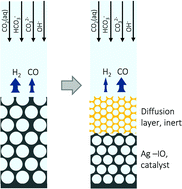
Mesostructured silver catalysts have shown a substantial rise in reaction selectivity for the conversion of CO2 into fuels with increasing thickness of ordered, inverse-opal films. The challenge lies in the optimization of the morphology to maximize the CO selectivity. We developed a 3D mass transport model utilizing the accurate electrode geometry to calculate local concentration distributions of CO2(aq), OH−, HCO3−, and CO32− by considering the buffer reactions in the electrolyte and modeling local catalytic surface reaction rates based on Butler–Volmer correlations. Validated with experimental data from the literature, the model predicted the suppression of the hydrogen evolution reaction with an inverse dependency on the hydroxide concentration and the promotion of the CO evolution reaction with a proportional dependency on the carbonate concentration. In order to increase the CO selectivity, we developed design guidelines that suggest high electrode roughness per film thickness, which translates to smaller pore size in practice. Further, the shallow pores of the electrode strongly reduced the overall CO selectivity as the mass transport to the bulk was non-limiting. We demonstrated that the introduction of an additional diffusion layer on top of the silver electrode can enhance the CO selectivity from as low as 39% to more than 90%.


 Please wait while we load your content...
Please wait while we load your content...