The balance of electric field and interfacial catalysis in promoting water dissociation in bipolar membranes†
Abstract
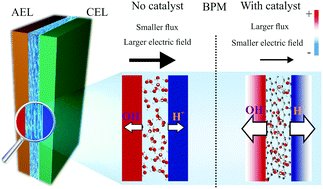
The lamination of a cation exchange layer (CEL) and an anion exchange layer (AEL) to form a hybrid bipolar membrane (BPM) can have several unique advantages over conventional monopolar ion exchange membranes in (photo-)electrolysis. Upon application of a reverse bias, the ordinarily slow water dissociation reaction at the AEL/CEL junction of the BPM is dramatically accelerated by the large electric field at the interface and by the presence of catalyst in the junction. Using electrochemical impedance spectroscopy (EIS), we have found a counterbalanced role of the electric field and the junction catalyst in accelerating water dissociation in a BPM. Experimental BPMs were prepared from a crosslinked AEL and a Nafion CEL, with a graphite oxide (GO) catalyst deposited at the junction using layer-by-layer (LBL) assembly. BPMs with an interfacial catalyst layer were found to have smaller electric fields at the interface compared to samples with no added catalyst. A comprehensive numerical simulation model showed that the damping of the electric field in BPMs with a catalyst layer is a result of a higher water dissociation product (H+/OH−) flux, which neutralizes the net charge density of the CEL and AEL. This conclusion is further substantiated by EIS studies of a high-performance 3D junction BPM that shows a low electric field due to the facile catalytic generation and transport of H+ and OH−. Numerical modeling of these effects in the BPM provides a prescription for designing membranes that function at lower overpotential. The potential drop across the synthetic BPM was lower than that of a commercial BPM by more than 200 mV at >100 mA cm−2 reverse bias current density, with the two membranes having similar long-term stability.


 Please wait while we load your content...
Please wait while we load your content...
