NaCrO2 cathode for high-rate sodium-ion batteries†
Abstract
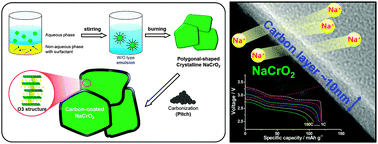
Sodium-ion batteries offer a potential alternative or complementary system to lithium-ion batteries, which are widely used in many applications. For this purpose, layered O3-type NaCrO2 for use as a cathode material in sodium-ion batteries was synthesized via an emulsion-drying method. The produced NaCrO2 was modified using pitch as a carbon source and the products were tested in half and full cells using a NaPF6-based non-aqueous electrolyte solution. The carbon-coated NaCrO2 cathode material exhibits excellent capacity retention with superior rate capability up to a rate of 150 C (99 mA h (g oxide)−1), which corresponds to full discharge during 27 s. The surface conducting carbon layer plays a critically important role in the excellent performance of this cathode material. We confirmed the reaction process with sodium using X-ray diffraction and X-ray absorption spectroscopy. Thermal analysis using time-resolved X-ray diffraction also demonstrated the structural stability of carbon-coated Na0.5CrO2. Due to the excellent performance of the cathode material described herein, this study has the potential to promote the accelerated development of sodium-ion batteries for a large number of applications.

 Please wait while we load your content...
Please wait while we load your content...