Rational design of a metal–organic framework host for sulfur storage in fast, long-cycle Li–S batteries†
Abstract
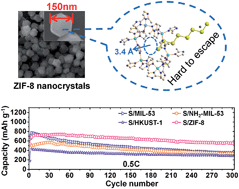
Unlike an intercalation cathode, which has an intrinsic host structure made of redox metal sites allowing the transport of Li+/e−, sulfur as a conversion cathode requires an additional host to store and immobilize the mobile redox centers, polysulfides. Metal–organic frameworks (MOFs) as a class of highly porous and well-defined crystalline materials are a promising platform to search for an effective host through rational design. With the appropriate selection of an electrolyte and a cutoff voltage range, sulfur stored in an appropriate MOF host can take advantage of both intercalation (fast and stable) and conversion (high energy density) cathodes. Herein, we describe a fast cathode with long cycle life based on sulfur and ZIF-8 nanocrystals. With 30 wt% sulfur loading in the electrode, it achieves remarkable discharge capacities of 1055 mA h g−1 (based on sulfur) at 0.1 C and 710 mA h g−1 at 1 C. The decay over 300 cycles at 0.5 C is 0.08% per cycle, prominent in long-cycle Li–S batteries. By comparing with another three distinct MOFs, MIL-53 (Al), NH2-MIL-53 (Al) and HKUST-1, as well as two sets of ZIF-8 with particle sizes in the micrometer range, it reveals that (i) the small particle size of the MOF host is appreciable to achieve a high capacity and (ii) small apertures, associated with functionalities in the open framework that have affinity with the polysulfide anions, can help achieve a stable cycling. We believe that the findings are general and applicable for the rational design of new hosts for sulfur in other porous material families to produce more effective and stable Li–S batteries.

 Please wait while we load your content...
Please wait while we load your content...