Improvement of open-circuit voltage and photovoltaic properties of 2D-conjugated polymers by alkylthio substitution†
Abstract
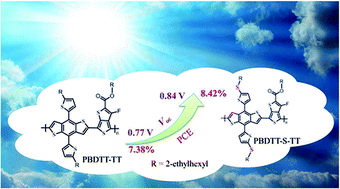
Low bandgap and two-dimensional (2D)-conjugated copolymers based on benzo[1,2-b:4,5-b′]dithiophene with conjugated thiophene side chains (BDTT) and thieno[3,4-b]thiophene with electron-withdrawing substituents (TT) are attractive high efficiency polymer donor materials in polymer solar cells (PSCs). In this work, we introduced an alkylthio substituent on the thiophene side chain in the polymer and synthesized a new low bandgap 2D-conjugated polymer PBDTT-S-TT. The alkylthio substituent increased the hole mobility of the polymer to 4.08 × 10−3 cm2 V−1 s−1 and down-shifted the HOMO energy level of the polymer by 0.11 eV with absorption of the polymer film red-shifted slightly. The PSCs based on PBDTT-S-TT as a donor and [6,6]-phenyl-C71-butyric acid methyl ester (PC70BM) as an acceptor without solvent additive treatment demonstrated a high open-circuit voltage (Voc) of 0.84 V, leading to a high power conversion efficiency (PCE) of 8.42%, under the illumination of AM 1.5 G 100 mW cm−2. For comparison, the Voc and PCE of the devices based on the corresponding parent polymer PBDTT-TT with the device optimization of 3% DIO additive treatment are 0.77 V and 7.38%, respectively. The enhanced Voc value of 0.84 V for the PSC based on PBDTT-S-TT should be benefited from the down-shifted HOMO energy level of the polymer. The results indicate that the alkylthio substitution is an effective way to further improve the photovoltaic performance of the 2D-conjugated polymer donor materials in PSCs.

 Please wait while we load your content...
Please wait while we load your content...