Cathodic shift of onset potential for water oxidation on a Ti4+ doped Fe2O3 photoanode by suppressing the back reaction†
Abstract
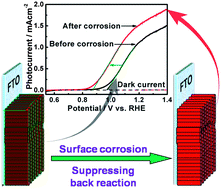
We present a surface corrosion method to shift the photocurrent onset potential cathodically for water oxidation on a Ti4+ doped Fe2O3 by about 100 mV. After the surface treatment, the doped hematite photoanodes showed a similar photocurrent onset potential to the lowest values obtained by loading with electrocatalysts or depositing functional over-layers. Moreover, the cathodic shift of the onset potential was preserved well even after a long operating time. The results indicated the effectiveness of this simple surface treatment. In order to make clear the reason for the onset potential shift, the doped hematite samples before and after surface corrosion were investigated by SEM, X-ray photoelectron spectroscopy (XPS), inductively coupled plasma mass spectrometry (ICP-MS), photoluminescence spectroscopy (PL), electrochemical impedance spectroscopy (EIS), Mott–Schottky and so on. Based on the experimental evidence, we proposed a new mechanism for the onset potential shift. The cathodic shift of the onset potential was due to decreasing the back reaction, but not accelerating water oxidation kinetics, passivating surface states or ions adsorption as reported in the previous studies. This strategy of suppressing the back reaction can offer a reference to reduce the overpotential for other photoelectrodes.

 Please wait while we load your content...
Please wait while we load your content...