The road from animal electricity to green energy: combining experiment and theory in electrocatalysis
Abstract
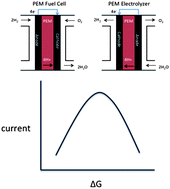
Advances in the field of electrocatalysis over the past several decades have been driven both by improvements in fundamental techniques for probing the solid–liquid electrochemical interface and by the technological imperative to develop enhanced low temperature electrocatalytic devices. In this review, we describe how a synergistic interaction between fundamental science and technological progress has resulted in both the emergence of greatly enhanced understanding of electrocatalytic systems and the development of practically improved electrocatalysts. Since it is not possible to summarize in detail all relevant developments in this broad field in such a brief space, we focus selectively on the early historical development of the field and on the use of trends-based analyses to describe the properties of electrocatalytic materials in terms of relatively simple catalytic properties, or descriptors. We begin by discussing aspects of the historical development of “reversible fuel cells” in acidic media, including topics relevant both to fuel cells, wherein hydrogen and oxygen are converted to water with concomitant production of electricity (electrons), and to electrolyzers, wherein electrons are used to initiate water splitting to yield hydrogen and oxygen. We then show how this development has stimulated the development of in situ and ex situ surface sensitive probes and spectroscopic methods capable of elucidating fundamental (atomic-/molecular-level) chemical and electronic properties of electrode–electrolyte interfaces. We further discuss how enhanced computational approaches, that can accurately calculate covalent bonding interactions in these systems, have contributed to the growth of a synergistic experimental/computational approach to electrochemical surface science that has resulted in a highly successful paradigm for the understanding of reactivity trends across a space of different metals, alloys, and metal oxides; this work, in turn, has spurred the development of alternative energy systems for efficient conversion and storage of chemical energy. We conclude with a discussion of some further needs for methodological developments and future research directions.

 Please wait while we load your content...
Please wait while we load your content...