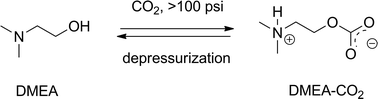
Anhydrous DMEA, DEEA and DIPEA are found to absorb carbon dioxide under pressure via chemical binding and physical absorption. The chemical CO2-bound derivatives of these materials are zwitterionic alkylcarbonate salts which are characterized by high-pressure 13C NMR. DMEA, DEEA and DIPEA absorb 20 wt.%, 17 wt.% and 16 wt.% carbon dioxide, respectively, at 300 psig (20.6 ATM). An increasing chemical carbon dioxide uptake capacity trend of DMEA > DEEA > DIPEA is observed while the physical CO2 absorption trend is DIPEA > DEEA > DMEA. DMEA captures up to 45 mole % (20 wt.%) of CO2 at 500 psig via both chemical binding and physical absorption. The amount of chemically bound and physically absorbed CO2 is directly linked to the CO2 pressure over the liquid. The zwitterion DMEA-CO2 regenerates CO2 and DMEA upon depressurization, allowing for an economical pressure swing regeneration rather than thermal regeneration. DMEA absorbs/releases CO2 repeatedly with no decline in capacity.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...